What is an Atom?
Despite draping themselves in bed sheets for every occasion, the Ancient Greeks were brilliant enough to conceive of atoms as building blocks of the universe. Without a whiff of technology at his disposal, Leucippus realised that all matter is composed of tiny bits. He named them atomos, which means indivisible.
This was a great starter hypothesis, even if the name turned out to be a total misnomer. Science has since brought us to the more robust conclusion that atoms are indeed divisible. What's more, all kinds of quantum quirks lurk within...
What Are Atoms?
As the building blocks of matter, atoms are substantially made of positively-charged protons and neutrally-charged neutrons.
Much smaller, negatively-charged electrons zap around them, balancing the overall electrical charge of the atom.
In 1913, Niels Bohr gave us a schematic model of atomic structure. It reveals these three core components to help explain how atoms work.
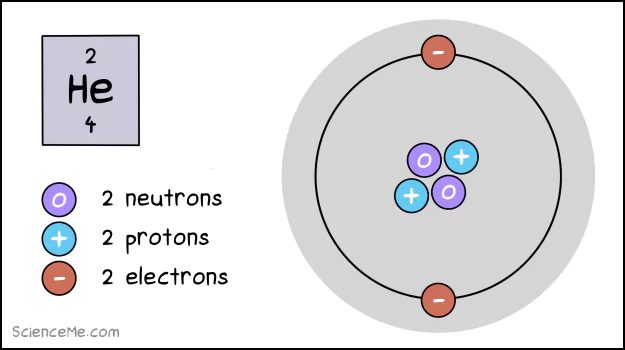
A helium atom (He) has 2 electrons, 2 protons, and 2 neutrons.
The number of protons in an atom tell us which element we're looking at, recorded as the atomic number.
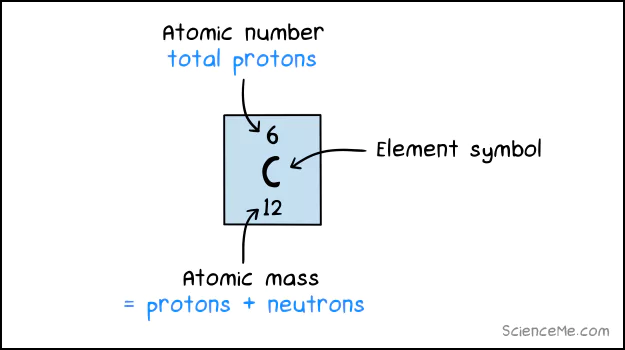
An atom of carbon (C) has 6 protons and 6 neutrons, giving it an atomic mass of 12 amu.
The lightest element in the universe is hydrogen (H) with one proton, while the heaviest naturally-occurring element is uranium (U) with 92 protons.
Physicists have gone and produced 24 heavier man-made elements in nuclear reactors and particle accelerators. They're named after famous scientists like the awkward-sounding rutherfordium (Rf) and einsteinium (Es).
The characters are laid out in the iconic Periodic Table of Elements, invented by Dmitri Mendeleev in 1869, who left gaps for elements yet to be discovered. Here's a modern version.
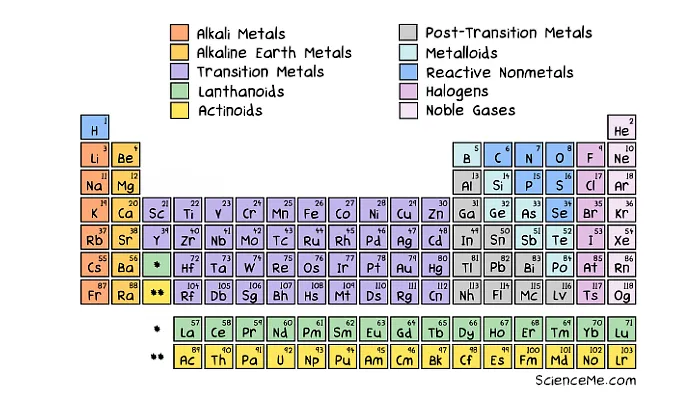
The Periodic Table of Elements reveals shared properties.
The arrangement may look odd, but it reveals key information about the number of electron orbits, also known as energy shells or energy levels. The electrons in the outermost shell dictate how atoms bond together to form solids, liquids, and gases.
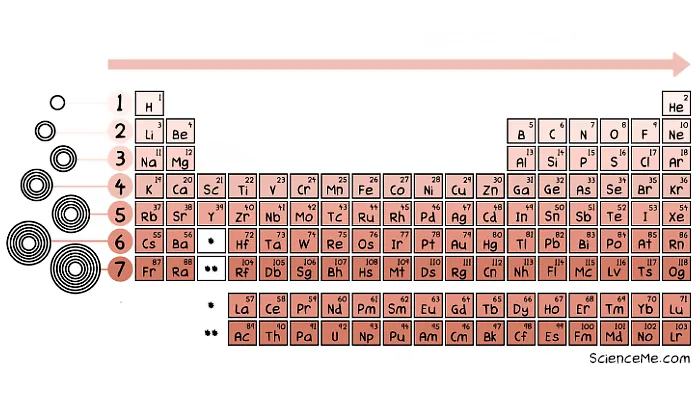
The Periodic Table also reveals the energy levels of each atomic species in its default state.
Electrons, Energy Shells, and Bonds
Electrons are super-tiny and super-sneaky. We're going to look at their behaviour in atomic orbits, but it's worth noting that they can also roam free to serve as the primary carrier of electricity.
In atoms, electrons move around in designated energy shells. The first energy shell (K) holds up to 2 electrons, the second shell (L) holds 8, the third shell (M) holds 18, the fourth shell (N) holds 32, and so on.
These energy levels are further divided into subshells (called s, p, d, and f) but, hey, this is Atoms 101 so let's not go there yet.
It's the behaviour of electrons in the outermost shell that determines how atoms bond to form molecules.
For instance, oxygen atoms can bond together to produce a molecule of oxygen (O2), while hydrogen and oxygen can bond to make water (H2O). Both are very handy molecules for all animal life on Earth, so let's find out how they occur.
1. Covalent Bonds
Covalent bonding is the most common way for atoms to get together. It involves sharing electrons between their outermost shells.
For instance, oxygen has six electrons in its outermost shell, with the capacity for two more. It's like that incomplete feeling you get when only six guests turn up to your dinner table reservation of eight.
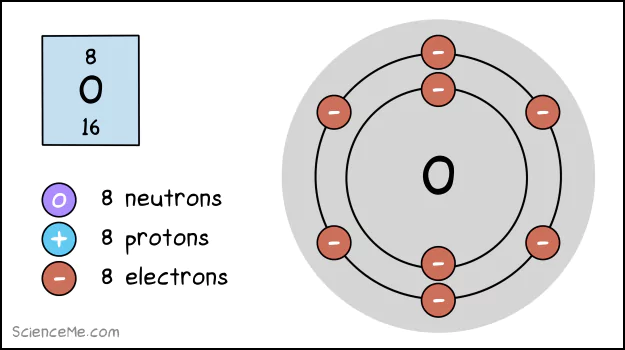
An atom of oxygen has 8 electrons, 8 protons, and 8 neutrons.
The easiest way to resolve this is to invite 2 more electrons to the table. But where can they come from?
Two hydrogen atoms step up. Hydrogen has only 1 electron in its outer shell. Just one, lonely diner who really wants a date.
Now, this is where my dinner table analogy gets weird. Because the solution is to have the waiters push their tables closer together—and have electron diners hop continuously back and forth.
Now it's more like a chaotically organised speed dating event.
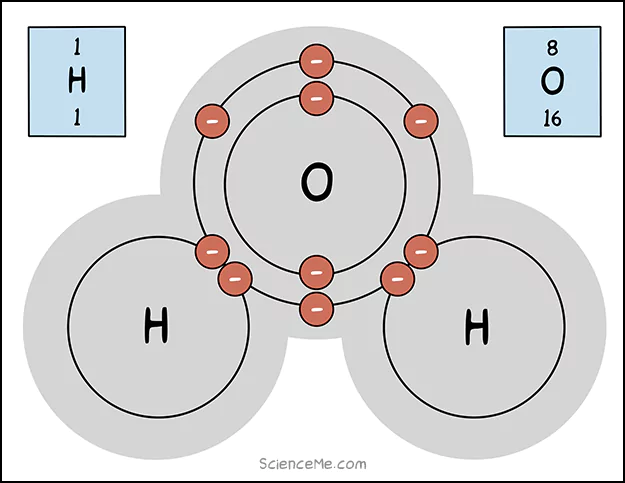
A water molecule is formed when an oxygen atom forms covalent bonds with two hydrogen atoms.
What's really happening here? The electrons zap around between the energy shells, sometimes completing oxygen with 8 outer electrons, and sometimes completing hydrogen with 2.
Incidentally, electrons seek to operate in pairs with opposite spins. Having the same negative charge also sees these pairs repelled by each other, doubling down on the saying that opposites attract. This fun feature of electron behaviour forces molecules to take up distinct and predictable shapes.
Molecules have predictable shapes owing to the repulsion of electron pairs.
Covalent bonds create reversible alliances between most elements in the universe. Water is one of the universe's best ideas so far, making up 90% of your body and the medium for yet more chemical reactions.
2. Ionic Bonds
Instead of politely sharing electrons, some atoms outright steal them. This is fine though, because their partners are willing donors. There's no foul play in physics.
To make table salt, for instance, atoms of sodium (Na) and chlorine (Cl) come together as sodium chloride (NaCl).
With 7 electrons in its outer shell, chlorine is on the lookout for an extra electron. It sees this opportunity in sodium.
Similarly, sodium has just 1 electron in its outer shell which also wants to buddy-up. So it jumps to chlorine. The sodium atom drops its empty energy shell, while the chlorine atom feels complete with all its electrons paired up.
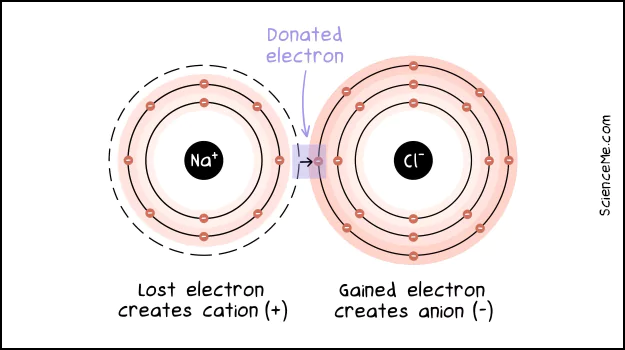
Ionic bonding involves electron donation to create positive and negative ions.
This is all well and good, but what's keeping sodium and chlorine together if they're not sharing electrons? It comes down to the new attraction between the residual charges of the atoms.
When sodium drops to 10 electrons, the 11 protons in the nucleus leave the atom with an overall positive charge of +1.
Likewise, chlorine upgrades to 18 electrons, yet only has 17 protons in its nucleus. It's left with a negative charge of -1.
Due to these unbalanced electrical charges, the atoms are now called an ions. The ionic attraction between the oppositely-charged atoms gives us a tasty molecule of salt.
What Are Isotopes?
We've looked at the effect of varying protons (giving us elements) and varying electrons (giving us molecules). But what happens when atoms have varying numbers of neutrons?
Now we get isotopes: the same element but now in new and improved flavours due to the unusual number of neutrons.
Isotopes can be stable or unstable (radioactive), and can form naturally through cosmic ray bombardment or radioactive decay, as well as artificially in a nuclear reactor.
For instance, radioactive C-14 isotopes are formed in the upper atmosphere as a result of cosmogenic radiation producing extra neutrons. This carbon-14 decays over time, with a half-life of 5,730 years, which is useful in carbon dating where the age of fossils can be estimated based on the ratio of carbon isotopes.

Carbon has three isotopes—C-12, C-13, and C-14—determined by the number of neutrons.
What Do Atoms Really Look Like?
Earlier we imagined electrons orbiting the nucleus of an atom, the same way we envision the planets orbiting the Sun. While based on Bohr's classic model of atomic structure, we must now accept that this model is hideously oversimplified. Sorry about that.
Electrons actually pop in and out of existence somewhere within their designated three-dimensional energy shells.
The electron orbits aren't flat on a plane. And their paths aren't continuous. It's much, much messier.
Fortunately, Bohr also figured out that the location of electrons can be roughly predicted using maths. But why only roughly, Goddammit? I need to know where the electrons are at all times.
One of the weird things about quantum mechanics is that we can only know the probability of subatomic particles being in any particular place at any particular moment. Dive into this conundrum more with the Double Slit experiment.
This uncertainty produces a probability density showing where electrons are most likely lurking at any moment, giving us an overall sense of the shape of an atom.

Visualising the probability density of hydrogen electron orbitals.
But this is still a conceptual representation. Is it even possible to see an atom, for real?
It is. Check out this long-exposure photo of a positively charged strontium ion.

Oh, you can't see it? Let's zoom in.

There it is... in among all those other atoms.
The strontium ion is illuminated with a blue-violet light and held motionless by an electric field. We're no better off understanding what an atom looks like, but it's still a cool photo. And it was taken with a digital SLR camera.
If that doesn't float your boat, take a look at this picture of a hydrogen atom. It's under attack by laser pulses which force its one and only electron to try to escape along direct and indirect trajectories. The phase difference between these trajectories produces an interference pattern.

Hydrogen Atom Under Magnification
It's the best view nature will let us have. The electron isn't in all these places at once (or is it?) but we do know it's somewhere in that turquoise region.
Of course, you can see what atoms look like any time you want, just at a terrible resolution. As you read these words, what you see to the best of your optical capacity is the accumulation of countless atoms. They've been staring at you in the face all along.
All Articles

16 Giant Leaps in Animal Evolution
An evolutionary sprint from the first clumps of cells wobbling about in the ocean to the hairy, noisy bipeds we are today.

15 Popular Science Books
Reading is like getting into another person's brain pool and swimming around in their knowledge. There is only one rule. DON'T PEE IN THE POOL.

Are We More Than Biological Machines?
If thoughts and sensations are cities, then consciousness is the interconnected network of highways that connect them.

Helium Could Save Our Planet
Huge helium reserves just beneath the Moon's surface could supply nuclear fusion reactors on Earth, offering a clean energy source for the 21st century.

Neuralink and You
Take a coin-sized panel of microchips, a battery, and 3,000 electrodes distributed along neural threads thinner than human hair. This is a Link. And it goes inside your skull.

How Was Stonehenge Built?
In the 5,000 years since it was built, Stonehenge has been eroded by weather, sunken by earthworms, and picked at by handsy tourists.

How Does DNA Work?
DNA isn't just a blueprint for foetal growth—you're expressing DNA right now to produce life-sustaining proteins like insulin, cortisol, and oxytocin.

Who Owns Your Organs When You Die?
It's not you—it's me. Besides a pseudo-compassionate breakup line, it's also the answer to this ethical minefield of a question.

12 Rules for Life Illustrated
12 Rules for Life is a psychological manifesto from a man who's making a giant existential omelette and breaking more than a few eggs in the process.

The Life of Elon Musk
So you have this dream of changing the world? For Elon Musk, this isn't enough. His ambitions are multiplanetary.





